Main page
About us
Sliding Bearings Consulting
Advertising Opportunities

to Metals
to Steel making
Macrosegregation in steel ingots
Dr. Dmitri Kopeliovich
Solidification of a liquid alloy is characterized by a phenomenon called segregation.
Segregation is a separation of impurities and alloying elements in different regions of solidified alloy.
Segregation is caused by the rejection of the solutes from a solidified alloy into the liquid phase. This rejection is a result of different solubility of impurities in liquid and solid phases at the equilibrium temperature.
Partition of solutes between the dendrite arms and interdendritic liquid is called Microsegregation.
Causes of macrosegregation
Microsegregation is the basic cause of macrosegregation. Microsegregation results in formation of numerous small volumes of liquid enriched with the solute. The liquid is surrounded by the solid dendrites having low solute concentration (solute-poor).
Transfer of the solute-rich liquid phase or solute-poor solid phase to large distances over the solidifying mass results in enrichment (positive macrosegregation) or impoverishment (negative macrosegregation) of particular zones of the ingot/casting with the solute.
The macro movements of the liquid and solid phases may be caused by the following effects:
Shrinkage of solidifying alloy causes suction of the liquid phase through the interdendritic structure. The moving liquid feeds the shrinking metal.
- Buoyancy forces
Liquid phase may move as a result of a gradient of the density caused by gradients of temperature and solute concentration.
- Zone melting effect
A separate volume of the liquid phase having different temperatures at its opposite sides may “move” in the direction of the higher temperature. The volume solidifies at the colder side and melts solid phase at the hotter side like in zone melting.
- CO bubbles
CO bubbles form in solidifying steel as a result of either low content of metallic deoxidizers in rimmed steel or Ladle-to-mold degassing in vacuum.
Floating gas bubbles cause movement of the solidifying alloy.
- External forces
Metal movement may be caused by the energy of the stream poured into the mold from the ladle. Solidified metal may be also forced by electro-magnetic field, mechanical stirrers, vibration.
- Sedimentation of solid phase
Solid crystals homogeneously nucleated in liquid phase and fragments of broken or melted off dendrites settle down (they are commonly heavier than liquid.
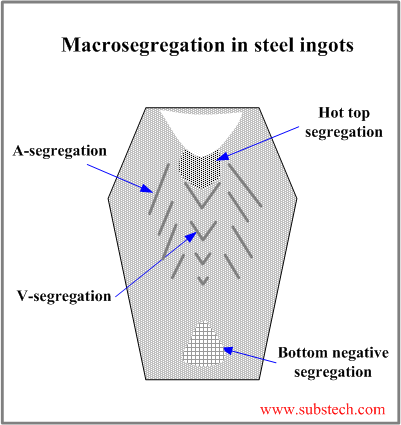
Macrosegregation zones in steel ingot
Development of macrosegregation zones in a steel ingot and their locations are associated with the ingot grain structure.
- Bottom negative segregation
Bottom negative segregation is a result of low solute concentration in the crystals formed in the early stage of solidification and comprising bottom cone. The bottom cone is a mixture of small equiaxed garins grown as a result of the contact with a bottom of a cold metallic mold and crystals and crystals fragments, which sedimintate from other ingot zones.
- V-segregation
The central zone of ingot is enriched with solute rejected by the solidification front progressing from the mold wall to its center. The central zone consists of large equiaxed grains, which settle down to the V-shaped solidification front. The residual liquid surrounding the large equiaxed grains is solute-rich and it forms V-segregates when solidifies.
- A-segregation
A-segregates (freckles) form in the Zone of columnar grains at the regions with structure characterized by the transition from the columnar grains to large equiaxed grains. A-segrgates present channels enriched by sulfur, carbon, phosphorus and other impurities.
- Hot top segregation
Hot top segregation zone is located in the top central ingot region below the shrinkage cavity. Hot top segregation is formed at the final solidification stage from the residual liquid enriched by the solutes as a result of microsegregation (rejection by solidifying dendrites) followed by penetration of the liquid through the dendrite skeleton.
to top
Related internal links
to Metals
to Steel making