Main page
About us
Sliding Bearings Consulting
Advertising Opportunities

to Metals
to Steel making
Basic Oxygen Furnace (BOF)
Dr. Dmitri Kopeliovich
Basic Oxygen Furnace (BOF) is a steel making furnace, in which molten pig iron and steel scrap convert into steel due to oxidizing action of oxygen blown into the melt under a basic slag.
The Basic Oxygen Process (Basic Oxygen Furnace, Basic Oxygen Steelmaking, Basic Oxygen Converter) is the most powerful and effective steel making method. About 67% of the crude steel in the world is made in the Basic Oxygen Furnaces (BOF).
Structure of a Basic Oxygen Furnace
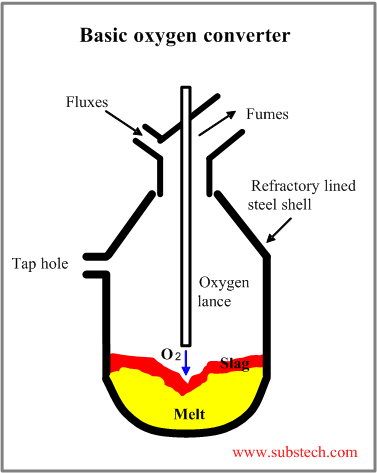
The scheme of a Basic Oxygen Furnace (BOF) is presented in the picture.
Typical basic oxygen furnace has a vertical vessel lined with refractory lining.
Only 8-12% of the furnace volume is filled with the treated molten metal. The bath depth is about 4-6.5 ft (1.2-1.9 m). The ratio between the height and diameter of the furnace is 1.2-1.5. The typical capacity of the Basic Oxygen Furnace is 250-400 t.
The vessel consists of three parts: spherical bottom, cylindrical shell and upper cone. The vessel is attached to a supporting ring equipped with trunnions.
The supporting ring provides stable position of the vessel during oxygen blowing.
The converter is capable to rotate about its horizontal axis on trunnions driven by electric motors. This rotation (tilting) is necessary for charging raw materials, sampling the melt and pouring the steel out of the converter.
The top blown basic oxygen furnace is equipped with the water cooled oxygen for blowing oxygen into the melt through 4-6 nozzles. Oxygen flow commonly reaches 200-280 ft3/(min*t) (6-8 m3/(min*t)). The oxygen pressure is 150-220 psi (1-1.5 MPa). Service life of oxygen lance is about 400 heats.
The bottom blown basic oxygen furnace is equipped with 15-20 tuyeres for injection of oxygen (or oxygen with lime powder). The tuyeres are cooled by either hydrocarbon gas (propane, methane) or oil supplied to the outer jacket of the tube.
to top
Refractory lining of a Basic Oxygen Furnace
The refractory lining of basic oxygen furnaces work in severe conditions of high temperature and oxidizing atmosphere. The lining wear is fastest in the zone of contact with the oxidizing slag (slag line).
Refractory bricks for lining basic oxygen furnaces are made of either resin bonded magnesite or tar bonded mixtures of magnesite (MgO) and burnt lime (CaO). The bonding material (resin, tar) is coked and turns into a carbon network binding the refractory grains, preventing wetting by the slag and protecting the lining the from chemical attack of the molten metal.
The following measures allows to prolong the service life of the lining:
- Control of the content of aggressive oxidizing oxide FeO in the slags at low level.
- Addition of MgO to the slags.
- Performing “slag splashes” - projecting residual magnesia saturated slag to the lining walls by Nitrogen blown through the lance.
- Repair the damaged zones of the lining by gunning refractory materials.
Properly maintained lining may serve 20000 heats.
to top
Chemical and physical processes in a Basic Oxygen Furnace
The basic oxygen furnace uses no additional fuel. The pig iron impurities (carbon, silicon, manganese and phosphorous) serve as fuel. Iron and its impurities oxidize evolving heat necessary for the process.
Oxidation of the molten metal and the slag is complicated process proceeding in several stages and occurring simultaneously on the boundaries between different phases (gas-metal, gas-slag, slag-metal). Finally the reactions may be presented as follows:
(square brackets [ ] - signify solution in steel, round brackets ( ) - in slag, curly brackets {} - in gas)
1/2{O2} = [O]
[Fe] + 1/2{O2} = (FeO)
[Si] + {O2} = (SiO2)
[Mn] + 1/2{O2} = (MnO)
2[P] + 5/2{O2} = (P2O5)
[C] + 1/2{O2} = {CO}
{CO} + 1/2{O2} = {CO2}
Most oxides are absorbed by the slag.
Gaseous products CO and CO2 are transferred to the atmosphere and removed by the exhausting system. Oxidizing potential of the atmosphere is characterized by the post-combustion ratio: {CO2}/({CO2}+{CO}).
Basic Oxygen Process has limiting ability for desulfurization. The most popular method of desulfurization is removal of sulfur from molten steel to the basic reducing slag. However the slag formed in the Basic Oxygen Furnace is oxidizing (not reducing) therefore maximum value of distribution coefficient of sulfur in the process is about 10, which may be achieved in the slags containing high concentrations of CaO.
to top
Operation of a Basic Oxygen Furnace
- Charging steel scrap (25-30% of the total charge weight).
- Pouring molten pig iron from blast furnace.
- Charging fluxes.
- Starting oxygen blowing. Duration of the blowing is about 20 min.
- Sampling. Temperature measurements (by disposable thermocouple) and taking samples for chemical analysis are made through the upper cone in tilted position of the furnace.
- Tapping - pouring the steel to a ladle. Special devices (plugs, slag detectors) prevent penetration (carry-over) of the slag into the ladle.
- De-slagging - pouring the residual slag into the slag pot. The furnace is turned upside down in the direction opposite to the tapping hole.
The Basic Oxygen Furnace has a capacity up to 400 t and production cycle (tap-to-tap) of about 40 min.
to top
Related internal links
to Metals
to Steel making